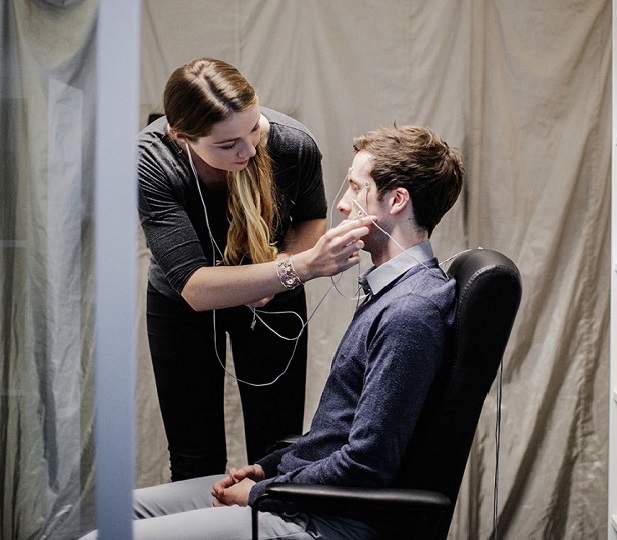
Lenire Tinnitus Clinical Trial Takeaways
95%
95% of compliant TENT-A2 tinnitus clinical trial patients reported improvement when evaluated after 12 weeks.2
91%
91% of TENT-A2 participants saw sustained improvement in tinnitus severity when evaluated at 12-months post-treatment.2
600+
Lenire has shown to provide clinically significant relief from tinnitus in clinical trials involving more than 600 participants.4
What are Clinical Trials?
Clinical trials aim to develop knowledge in a specific field. Trials help us understand and validate the effectiveness of new treatments.
Trials are regarded as the most responsible way of developing new medical interventions from research to public use. During trials, patients receive medical interventions (i.e. devices or drugs) and are observed by experts to monitor reactions.
Example: Lead by a Principal Investigator and an expert research team, 326 patients used Lenire for 12-weeks during TENT-A1 (Lenire Tinnitus Clinical Trial 1). 1

Why are Clinical Trials Important?
Ensuring Public Safety and Effectiveness

Effectiveness
Clinical trials examine the new interventions to test if they are effective at curing / soothing a condition or preventing it from worsening.

Safety
Clinical trials rigorously examine the safety of new intervention to understand if any risks outweigh benefits.
Unique Side Effects
Trials examine new interventions to identify unique factors that may cause adverse side effects on a patient.
Peer Reviewed & Real World Research
Publication of clinical studies validates research and allows healthcare professionals to trust scientific research in pursuit of adopting new medical practices.
During peer review, independent experts evaluate and validate methodologies and claims to determine if the research is worthy of publication in scientific journals.

TENT-A2 Research Paper:
Different bimodal neuromodulation settings reduce tinnitus symptoms in a large randomized trial
Published on: 30/06/2022 Corresponding author: Hubert Lim Download.pdf (3.97 MB)
TENT-A2 Protocol Paper:
Noninvasive Bimodal Neuromodulation for the Treatment of Tinnitus: Protocol for a Second Large-Scale Double-Blind Randomized Clinical Trial to Optimize Stimulation Parameters
Published on: 01/09/2019 Corresponding author: Hubert Lim Download.pdf (556.43 KB)
TENT-A1 Research Paper:
Bimodal neuromodulation combining sound and tongue stimulation reduces tinnitus symptoms in a large randomized clinical study (2020)
Published on: 07/10/2020 Corresponding author: Hubert Lim Download.pdf (1.68 MB)
TENT-A1 Supplementary Materials:
Supplementary Materials for Bimodal neuromodulation combining sound and tongue stimulation reduces tinnitus symptoms in a large randomized clinical study
Published on: 07/10/2020 Corresponding author: Hubert Lim Download.pdf (3.06 MB)
Retrospective Analysis of Real-World Patients:
Real-world clinical experience with bimodal neuromodulation for the
treatment of tinnitus.

TAVSS Research Paper:
An Investigation of Feasibility and Safety of Bi-Modal Stimulation for the Treatment of Tinnitus: An Open-Label Pilot Study
Published on: 04/02/2016 Corresponding author: Caroline Hamilton Download.pdf (117.62 KB)Lenire Tinnitus Clinical Trial Summaries
Treatment Evaluation of Neuromodulation for Tinnitus Stage 3 (TENT-A3)
Repeated Measure Controlled Trial
2022, 112 participants
Following TENT-A3, Lenire became the first and only FDA Approved device for the treatment of tinnitus. TENT-A3 results are currently being peer-reviewed for publication. TENT-A3 demonstrated that Lenire’s dual mode approach was more effective at treating moderate or worse tinnitus than sound-alone.
Treatment Evaluation of Neuromodulation for Tinnitus Stage 2 (TENT-A2)
Double-Blinded Randomized Trial
2019, 191 participants
Lenire’s second clinical trial, TENT-A2, optimised the Lenire’s settings to build off TENT-A1’s success.As a result, TENT-A2 demonstrated improved performance to TENT-A1.
Treatment Evaluation of Neuromodulation for Tinnitus Stage 1 (TENT-A1)
Double-Blinded Randomized Tinnitus Clinical Trial
2017, 326 participants
Landmark tinnitus clinical trial, TENT-A1, demonstrated that Lenire is safe and effective in reducing tinnitus in a large cross section of tinnitus patients.
Real World Evidence
Real World Clinical Setting Trial
2022, 204 Analysed Real World Cases
Conducted at the German Hearing Center (DHZ), this study demonstrated that Lenire can safely achieve a meaningful improvement for 85% of participants in a real-world clinical setting. Results were published in Brain Stimulation, February 2022.
Tinnitus Alleviation Via Sensory (TAVS) Stimulation
Safety and Feasibility Study
2015, 60 participants
The safety and feasibility study was designed to assess the safety of treatment prior to proceeding to large scale clinical trials.
Healthcare Professionals
Learn more about how adding Lenire to your tinnitus treatment options. Visit the Lenire for Healthcare Professionals page. Get in touch to learn more about tinnitus clinic trials.

Additional Papers and Case Studies
Read more about Lenire’s commitment to scientific solutions.
- 18/01/2024Tinnitus: A Dimensionally Segregated, yet Perceptually Integrated Heterogeneous Disorder
- 28/04/2023Using Lenire on a Patient with Acoustic Neuroma
- 28/02/2023Bimodal Neuromodulation Treatment for Tinnitus Positively Affects Various Aspects of Life
- 23/05/2022Usability of Bimodal Neuromodulation to Treat Tinnitus
- 04/08/2020www.scientificamerican.com – Scientific American’s coverage
- 24/04/2020www.newscientist.com – New Scientist’s coverage
- 01/03/2024Textbook of Tinnitus: Second Edition
1. TENT-A1 Conlon et al., Sci. Transl. Med. 12, eabb2830 (2020)
2. TENT-A2: Conlon et al., Different bimodal neuromodulation settings reduce tinnitus symptoms in a large randomized trial, Sci Rep.
3. TENT-A3: Clinical trial data in preparation for independent publication: clinicaltrials.gov/ct2/show/NCT05227365.
4. Aggregate: TENT-A1, TENT-A2, TENT-A3 aggregate clinical trial results.
Note 1: All statistics based on compliant patients. Patient compliance criteria is detailed in Lenire’s clinical trial design.
Note 2: TENT-A1/TENT-A2 were not considered by the United States of America FDA as part of Lenire’s FDA Grant and, as such, applies solely to outside the US.